 
However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner. The vast majority of its mass is located in a positively charged central nucleus, which is 10,000. Rutherford had made the startling discovery that most of the atom is empty space. It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. Bohr, one of the pioneers of quantum theory, had taken the atomic model presented a few years earlier by physicist Ernest Rutherford and given it a quantum twist. Pertama, jarak dan orbit yang ditekankan dalam model atom Bohr tidak sejalan dengan Prinsip Ketidakpastian Heisenberg, yakni jarak/radius tak bisa bersama dengan orbit. 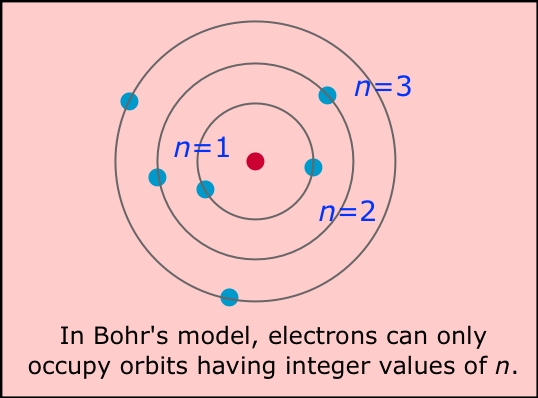
For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. The Bohr model of the atom, a radical departure from earlier, classical descriptions, was the first that incorporated quantum theory and was the predecessor of wholly quantum-mechanical models. Thus, the Bohr model for heavier atoms described electron shells. Bohr model, description of the structure of atoms proposed in 1913 by the Danish physicist Niels Bohr. The motion of the electrons in the Rutherford model was unstable because. The model was proposed by physicist Niels Bohr in 1913. In 1913, just two years after the Rutherford atomic model had been introduced, Danish physicist Niels Bohr, a student of Rutherford’s, proposed his quantized shell model of the atom (see Bohr model) to explain how electrons can have stable orbits around the nucleus. Once the level was full, additional electrons would be bumped up to the next level. The Bohr Model is a structural model of an atom. Bohr believed each electron orbit could only hold a set number of electrons. More electrons were required to cancel out the positive charge of all of these protons. In Bohr’s model, radius an of the orbit n is given by the formula an h2n2 0 / 2, where 0 is the electric constant. Heavier atoms contain more protons in the nucleus than the hydrogen atom. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
